Author: Bob Brensel | President, Pharmacist | ScriptWorks
Bob Brensel, RPh, earned his Pharmacy Degree at University of the Pacific in Stockton, California in 1980. Former California Pharmacists Association’s Award Winner for Recognition of Outstanding Achievement in Compounding Pharmacy. Read More →
Estimated Read Time: 10–12 minutes
Quick Summary:
Vaginal hormone creams may be considered when menopausal symptoms are primarily localized to vaginal tissue. Providers may evaluate several hormone options including:
- Estradiol (E2)
- Estriol (E3)
- Bi-Est (E2 + E3)
- Progesterone
- Testosterone
- DHEA
Localized therapy may be evaluated when symptoms such as vaginal dryness, irritation, or discomfort during intimacy are present.
Article Overview:
This article examines the clinical considerations surrounding a vaginal hormone cream commonly prescribed for menopause, particularly for patients with genitourinary syndrome of menopause (GSM) whose symptoms are primarily localized rather than systemic. It reviews the evidence behind the potential of low-dose vaginal hormone therapy, including observed effects on vaginal pH and epithelial maturation, and clarifies when localized therapies may be appropriate within a broader menopause care plan.
The discussion also outlines scenarios where adjustable strength, base selection, or combination therapy (e.g., estradiol, estriol, Bi-Est, progesterone, testosterone, or DHEA) may support individualized prescribing.
California Women in Menopause
California is home to millions of women navigating the menopausal transition, and many explore health options such as vaginal hormone cream for menopause when symptoms become disruptive. In fact, an estimated 5 million women in California are between the ages of 40 and 64 — the stage of life when menopause and perimenopause most commonly occur.

“California has about 5 million women ages 40–64, which is the typical age range for perimenopause and menopause.”
Source: CalHealth
Menopause can feel isolating — especially when symptoms affect intimacy, comfort, or daily confidence. But working with both medical practitioners and a reputable compounding pharmacy specializing in women’s hormone replacement, like ScriptWorks, provide patients with a personalized approach to hormone balance guiding them through each step of the way. Women do not have to go through this journey alone.
For many women, vaginal dryness, irritation, or discomfort during intimacy are among the most disruptive changes. When symptoms are primarily vaginal, localized hormone therapy may be considered — either alone or alongside a broader hormone evaluation.
Bioidentical hormone replacement therapy (BHRT) in the form of prescription vaginal hormone creams, may become part of a structured care plan developed with a healthcare practitioner for California women in menopause.
Why Do Patients Choose Bioidentical Hormone Replacement Therapy (BHRT)?
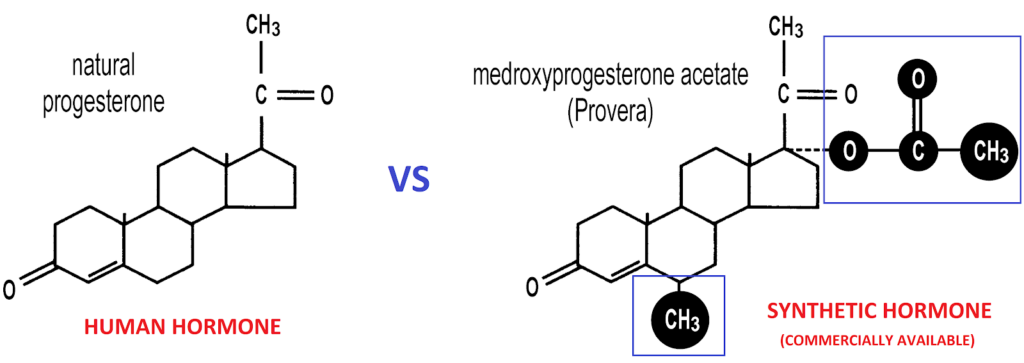
Bioidentical hormones are formulated to match the molecular structure of hormones produced by the body, while some commercially available hormone therapies contain synthetic or modified hormone compounds that differ structurally but are designed to act on the same hormone receptors.
Commercial Synthetic Hormones vs Compounded Bioidentical Hormones
Commercial vaginal estrogen products are available in fixed strengths and formats.
When appropriate, a prescriber may request a compounded vaginal preparation to better match an individual patient’s needs.
Commercial Products
- Fixed strength
- Fixed base
- Limited format options
- Standard excipient profile
Compounded Formulations
- Adjustable hormone strength (mg/g)
- Customizable base selection
- Option to combine ingredients when clinically appropriate
- Modified dispensing methods
- Tailored for ingredient sensitivities or tolerability concerns
Compounded medications are prepared for individual patients based on a prescription from a licensed healthcare provider. These customized formulations are not FDA-approved. They are created when commercially available options may not meet specific clinical needs.
ScriptWorks collaborates directly with the prescribing clinician to prepare the formulation exactly as specified, even providing insights to help practitioners make informed decisions.
What Is a Vaginal Hormone Cream?
A vaginal hormone cream is a prescription medication applied locally to vaginal tissue via vaginal cream applicator. It may contain estrogen or other hormone components and is used under provider supervision when symptoms such as dryness, irritation, or discomfort persist. Localized formulations focus on vaginal tissue directly and may be selected when symptoms are primarily genitourinary in nature.
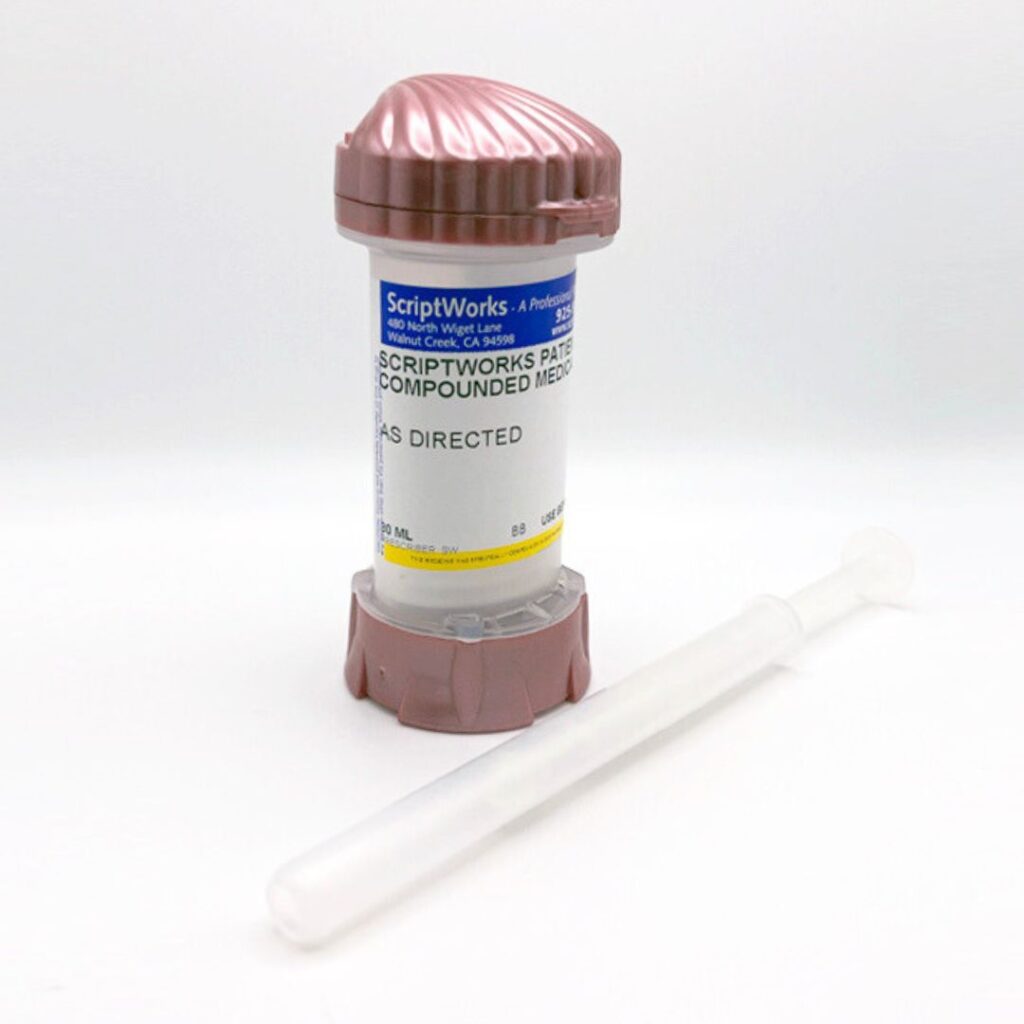
One method that may help support more precise and consistent application is the Topi-Click® PERL system vaginal applicator. This applicator is designed to deliver a measured amount of compounded cream with each click, which may help patients apply their medication more consistently. The device is designed with dosing accuracy and ease of use in mind, which may be helpful for individuals who prefer a straightforward and controlled application method.
The Topi-Click® PERL system may also support improved hygiene and convenience compared to traditional applicators, as the metered click system is designed to dispense a consistent amount of medication each time it is used. For some patients and providers, this type of delivery system may offer a practical option when using compounded vaginal creams.
It is important to note that with compounding, base selection matters. An anhydrous (water-free) vaginal gel interacts with natural vaginal moisture to form a micro-emulsion, designed to enhance localized contact with vaginal tissue.

Why May a Vaginal Cream Be Prescribed for Menopausal Women?
Vaginal administration has become more popular because the vaginal mucosa contains a rich blood supply and may allow hormones to interact directly with vaginal or nearby tissues.
Providers may consider vaginal hormone creams when:
- Vaginal dryness is the primary concern
- Discomfort during intimacy is present
- Urinary urgency or irritation accompanies vaginal changes
- Broader systemic symptoms are minimal
Types of Compounded Vaginal Hormone Creams for Menopause
When addressing vaginal symptoms are central to menopause care, providers may consider several localized compounded options.

Vaginal Estrogen Options
A review published in Obstetrics & Gynecology evaluated the role of vaginal estrogen therapy for GSM and discussed how localized estrogen preparations have been utilized for postmenopausal women experiencing symptoms such as vaginal dryness, irritation, and discomfort during intimacy.
Types of Vaginal Estrogen Used in Menopause Care
Vaginal Estradiol (E2)
Estradiol is one of the primary estrogens produced during the reproductive years. Vaginal estradiol formulations have been studied in postmenopausal women experiencing vulvovaginal symptoms associated with declining estrogen levels. Clinical research evaluating localized estradiol therapy has monitored changes in markers such as vaginal pH, epithelial maturation, and vaginal tissue health indicators over time.
Because estradiol interacts with estrogen receptors found in vaginal tissue, some healthcare providers may evaluate whether localized estradiol preparations may be appropriate when symptoms are primarily vaginal.
Healthcare providers may evaluate estradiol formulations in situations such as:
- More pronounced menopausal symptoms, including moderate to severe vaginal dryness or atrophy
- When both vaginal and broader menopausal symptoms are present, such as hot flashes or night sweats
- When stronger estrogen receptor activity may be desired based on the patient’s clinical evaluation
Vaginal Estriol (E3)
Estriol is another naturally occurring estrogen that has been explored in localized menopause therapies. Studies examining vaginal estriol preparations have monitored symptom reporting and vaginal tissue markers in postmenopausal women experiencing vaginal dryness or irritation.
Some providers may evaluate estriol-based formulations in situations such as:
- Symptoms that are primarily localized to vaginal tissue, such as dryness, irritation, or discomfort with intimacy
- When a lower-potency estrogen option may be preferred based on the patient’s clinical profile
- When localized vaginal therapy is the primary goal rather than systemic hormone therapy
- When combination estrogen therapy is being evaluated, such as Bi-Est formulations that include both estradiol and estriol
Vaginal Bi-Est (Estradiol + Estriol)
Bi-Est is a compounded formulation that combines estradiol and estriol, often in ratios such as 80:20 or 50:50.
Because these estrogens (Estradiol and Estriol) differ in potency and receptor activity, some clinicians evaluate combination formulations when designing individualized hormone therapy plans. Providers may consider vaginal Bi-Est formulations in situations such as:
- When a combination estrogen approach is preferred, incorporating both estradiol and estriol
- When individualized estrogen ratios may be desired, allowing prescribers to adjust the balance between estradiol and estriol
- When commercially available products do not offer the specific estrogen combination being evaluated
- When a compounded vaginal cream or gel is preferred for individualized dosing and formulation selection
Vaginal Progesterone Cream
Progesterone is a naturally occurring hormone that plays an important role in the female reproductive system and is commonly included in hormone therapy plans for certain postmenopausal women. In menopause care, progesterone is often discussed in the context of balancing estrogen exposure, particularly for women who retain their uterus.
Clinical literature has explored the pharmacologic characteristics and tissue exposure associated with vaginal progesterone therapy. Reviews published in journals such as Climacteric describe how vaginal progesterone may produce measurable hormone levels improvements while interacting with reproductive tissues through what researchers sometimes refer to as the “uterine first-pass effect,” a phenomenon in which medication delivered vaginally may reach uterine tissue directly through local circulation pathways.
Because of these pharmacologic properties, some healthcare providers evaluate whether vaginal progesterone formulations may be appropriate in specific clinical scenarios under medical supervision.
Vaginal Testosterone Cream
Testosterone is present in women in smaller amounts than in men and contributes to several physiologic processes, including aspects of sexual health and tissue function. As women transition through menopause, androgen levels may gradually decline. This change has led clinicians to explore whether localized testosterone therapy, such as vaginal testosterone cream, may play a role for certain patients experiencing vulvovaginal symptoms.
Clinical research has evaluated intravaginal testosterone therapy in postmenopausal women. In a randomized, double-blind, placebo-controlled trial published in the Journal of Clinical Endocrinology & Metabolism, investigators studied postmenopausal women experiencing vaginal symptoms associated with aromatase inhibitor therapy. Participants used intravaginal testosterone cream (300 micrograms per dose) for 26 weeks, and researchers monitored sexual health measures, vaginal symptoms, and hormone levels during the study period.
Investigators reported that women receiving intravaginal testosterone demonstrated changes in measures of sexual satisfaction, vaginal dryness, and dyspareunia compared with baseline assessments. Importantly, the study also monitored circulating hormone levels and observed no significant changes in serum sex steroid levels, suggesting limited systemic hormone changes during the observation period.
These findings contribute to the broader body of research exploring how localized androgen therapy may interact with vulvovaginal tissue. Because androgen receptors are present in vaginal tissue, some clinicians may evaluate whether vaginal testosterone cream could be appropriate in carefully selected patients when symptoms are primarily localized.
Vaginal DHEA Cream
Dehydroepiandrosterone (DHEA) is a hormone precursor produced by the adrenal glands that can be converted within the body into both estrogen and androgen hormones. Levels of DHEA gradually decline with age, and researchers have explored how localized DHEA therapy may interact with vaginal tissue in postmenopausal women.
Because vaginal tissues contain enzymes capable of converting DHEA into active hormones locally, some clinicians evaluate vaginal DHEA formulations when menopausal symptoms are primarily related to vulvovaginal tissue.
Healthcare providers may consider vaginal DHEA preparations in situations such as:
- Symptoms primarily affecting vaginal tissue, such as dryness, irritation, or discomfort during intimacy
- When localized hormone activity is being evaluated rather than systemic hormone therapy
- When a hormone precursor approach is preferred, allowing vaginal tissue to convert DHEA into small amounts of estrogen and androgen locally
- When patients are exploring alternatives to traditional estrogen therapy under medical supervision
Clinical research evaluating intravaginal DHEA has observed vaginal cell maturation, tissue health markers, and symptom reporting in postmenopausal women during therapy periods. These studies have contributed to ongoing clinical discussion regarding how localized DHEA therapy may interact with vulvovaginal tissue.
Safety and Medical Supervision
Hormone therapy decisions depend on individual medical history, symptom profile, and provider evaluation.
Important considerations may include:
- Presence or absence of a uterus
- Cardiovascular history
- Breast health history
- Thrombotic risk factors
- Concurrent medications
Ongoing provider supervision is essential. Compounded medications should only be used under the direction of a licensed healthcare provider.
Role of Hormone Testing in a BHRT Plan
Some providers incorporate laboratory testing as part of a structured hormone evaluation.
Testing methods may include blood or saliva analysis, depending on clinical judgment. Hormones that may be assessed include:
- Cortisol
- Estrogens
- Progesterone
- Testosterone
- DHEA
ScriptWorks offers at-home saliva collection kits for convenience. Our pharmacists go over the results with medical practitioners and their patients to cover recommendations, different approaches, and the next steps in their journey.
What to Expect When Starting a Compounded Vaginal Hormone Cream
1. Assessing Hormone Levels and Symptoms
Before a compounded vaginal hormone cream is prescribed, a healthcare provider typically evaluates both symptoms and hormone levels. This may include reviewing your health history, discussing vaginal or menopausal symptoms, and sometimes ordering laboratory testing to better understand hormone status.
Based on this assessment, the provider may determine whether a localized hormone preparation could be appropriate and decide on specific hormone types and strengths.
2. Determining the Appropriate Strength and Formula
Compounded vaginal creams allow prescribers to select the hormone concentration, base, and dosing schedule that may best align with a patient’s clinical picture. For example, a provider may prescribe a specific strength of estradiol or another hormone depending on the individual’s symptoms, lab findings, and overall care plan.
These formulations are prepared by a compounding pharmacy according to the provider’s prescription and may differ from patient to patient.
3. Early Changes After Starting
During the first few weeks of use, the vaginal tissues may gradually adjust to the presence of the hormone. Some individuals may begin to notice changes in vaginal comfort or moisture as the tissue environment adapts.
4. Expected Timeline
Experiences can vary from person to person, but providers often explain that changes may occur gradually:
- First few weeks: The body begins adjusting to the medication and local tissue exposure.
- 4–8 weeks: Some individuals may begin to notice more meaningful changes in vaginal comfort or dryness.
- Ongoing use: Providers may reassess symptoms and determine whether the formulation or dosing schedule should be adjusted.
Regular follow-up allows the provider to monitor progress and ensure the prescribed formulation remains appropriate.
5. Potential Benefits Patients May Notice
While responses differ for every individual, some people using localized vaginal hormone creams report improvements such as:
- Greater vaginal moisture
- Reduced dryness or irritation
- Improved comfort during daily activities or intimacy
- Overall improvement in vaginal tissue health markers
Because compounded medications are individualized, the experience can vary, and ongoing communication with a healthcare provider is important.
6. Follow-Up and Adjustments
Compounded therapy is often iterative. If symptoms change or new concerns arise, a provider may adjust the hormone strength, base, or dosing frequency. Follow-up visits help ensure the medication continues to align with the patient’s needs and goals.
Why California Women’s Health Practitioners Partner With ScriptWorks
ScriptWorks is a PCAB-accredited compounding pharmacy based in Walnut Creek, serving patients throughout California (statewide shipping).

SCRIPTWORKS
DELIVERS.
Your prescriptions arrive when you need them.
Get medications faster with ScriptWorks.
Weekday
Local Deliveries
Walnut Creek, Concord, Martinez, San Ramon, Danville, Alamo, Lafayette and Orinda.
USPS 1st Priority Mail
Overnight Mail Delivery
California (state-wide)
Since 2003, we have focused on personalized medication preparation — particularly in areas such as women’s hormone therapy, where standardized options may not always align with individual patient needs.

Menopause care and BHRT discussions begin in many types of clinical settings. We collaborate with licensed California practitioners across specialties, including:
- MDs, DOs, PAs, and NPs
- OB/GYN and women’s health specialists
- Hormone and menopause specialists
- Functional medicine providers
- Family practice and primary care clinicians
When hormone therapy is part of a patient’s care plan, we work directly with the prescribing clinician to prepare customized formulations aligned with their clinical direction.
Flexibility in Dosage Forms and More
Compounding allows providers to prescribe based on the patient in front of them when commercially available strengths may not align with specific clinical parameters. When a mass-produced, commercially made medication isn’t suitable due to strength limitations, ingredient sensitivities, or dosage form preferences, we work directly with the prescriber to prepare a customized formulation consistent with their clinical direction.
In addition to vaginal creams, compounding provides vast flexibility in dosage form and strength selection when clinically appropriate, including:
- Capsules
- Creams
- Gels
- Lozenges/troches
- Suppositories
- Ointments
- Oral liquids, solutions, or suspensions
- Nasal sprays
- Sublingual forms
- Script Tabs

This range supports individualized care while adhering to the quality controls and regulatory standards required of a California compounding pharmacy.
To support efficient workflow, ScriptWorks provides a secure Provider Portal and prescription order forms that allow clinicians to clearly specify strengths, bases, and dosage formats with ease. Our pharmacists are available for direct consultation when formulation clarification or collaboration is helpful.
California Women in Menopause: How to Get Started
For patients, the next step is simple: speak with your healthcare provider about your symptoms and ask whether localized hormone therapy or a broader BHRT plan may be appropriate. Ask your doctor to fill your prescription with ScriptWorks and your medication will be shipped directly to your door.
Menopause is personal, so the care approach must reflect that.
Thoughtful evaluation, individualized prescribing, and collaborative compounding allow menopause care to adapt — rather than remain fixed.
References
- Ballagh, S. A. (2005, May). Vaginal hormone therapy for urogenital and menopausal symptoms. Seminars in Reproductive Medicine, 23(2), 126–140. Link
- Davis, S. R., & Wahlin-Jacobsen, S. (2015). Testosterone in women—the clinical significance. The Lancet Diabetes & Endocrinology, 3(12), 980–992. Link
- Effectiveness and safety of vaginal suppositories for the treatment of vaginal atrophy in postmenopausal women: An open, non-controlled clinical trial. (2008, December 1). PubMed. Link
- Merlino, L., Ciminello, E., Volpicelli, A. I., Tillier, S., Pasquali, M. F., Dominoni, M., Gardella, B., Senatori, R., Dionisi, B., & Piccioni, M. G. (2024). Evaluation of the effectiveness of combined treatment with intravaginal diazepam and pelvic floor rehabilitation in patients with vulvodynia by ultrasound monitoring of biometric parameters of pelvic muscles: A pilot study. Diseases, 12(8), 174. Link
- Mitchell, C. M., Reed, S. D., Diem, S., Larson, J. C., Newton, K. M., Ensrud, K. E., et al. (2018). Efficacy of vaginal estradiol or vaginal moisturizer vs placebo for treating postmenopausal vulvovaginal symptoms: A randomized clinical trial. JAMA Internal Medicine, 178(5), 681–690. Link
- Mueck, A. O., Ruan, X., Prasauskas, V., Grob, P., & Ortmann, O. (2018). Treatment of vaginal atrophy with estriol and lactobacilli combination: A clinical review. Climacteric, 21(2), 140–147. Link
- Rueda, C., Osorio, A. M., Avellaneda, A. C., Pinzón, C. E., & Restrepo, O. I. (2017). The efficacy and safety of estriol to treat vulvovaginal atrophy in postmenopausal women: A systematic literature review. Climacteric, 20(4), 321–330. Link
- Santen, R. J. (2015). Vaginal administration of estradiol: effects of dose, preparation and timing on plasma estradiol levels. Climacteric, 18(2), 121–134. Link
- Stute, P., Neulen, J., & Wildt, L. (2016). The impact of micronized progesterone on the endometrium: A systematic review. Climacteric, 19(4), 316–328. Link
- Tanmahasamut, P., Jirasawas, T., Laiwejpithaya, S., Areeswate, C., Dangrat, C., & Silprasit, K. (2020). Effect of estradiol vaginal gel on vaginal atrophy in postmenopausal women: A randomized double-blind controlled trial. Journal of Obstetrics and Gynaecology Research, 46(8), 1425–1435. Link
- Traish, A. M., Kang, H. P., Saad, F., & Guay, A. T. (2011). Dehydroepiandrosterone (DHEA)—A precursor steroid or an active hormone in human physiology (CME). The Journal of Sexual Medicine, 8(11), 2960–2982. Link